|
Here are some different ways to draw aluminium oxide (Al 2 O 3). Example: Sodium chloride, Magnesium oxide, calcium chloride, etc., show this type of bonding. Nature tends to neutralize charges, so ions of opposite charge tend to attract to make ionic compounds. would be ionic from the position of the elements in the periodic table. An atom of sodium (Na) donates one of its electrons to an atom of chlorine (Cl) in a chemical reaction, and the resulting positive ion (Na +) and negative ion (Cl ) form a stable ionic compound (sodium chloride common table salt) based on this ionic bond. They are: Ionic bond: The electrostatic force of attraction which holds together ions of combining atoms formed by the complete transfer of one or more electrons from the electropositive to electronegative atom is called an ionic bond. 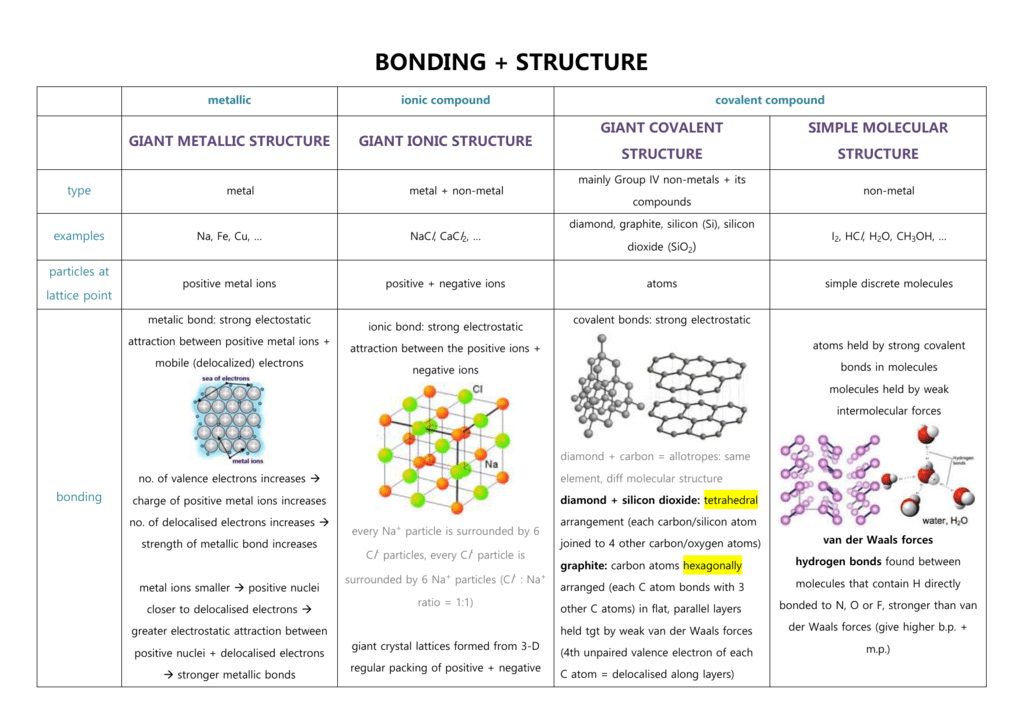
What happens when the charges on the ions are not equal in magnitude? In aluminium oxide the charge on the positive metal ions is 3 + while the charge on the negative oxide ions is 2 -. IB Chemistry standard level notes on ionic bonding. In magnesium oxide, the charges on the metal and non-metal ions are equal and opposite. Youll receive 2 points for each correctly built molecule. Ionic bonds occur between metals and non-metals on the periodic table. Magnesium oxide is not soluble in water because the attraction between the polar water molecules and the ions is not strong enough to break the ionic bonds between the magnesium and oxygen ions. In this game, your challenge is to create a compound by combining individual ions. In modern language, the central idea of an ionic bond is that electrons (one or.You can find the ionic charge of these ions using the Periodic Table. It forms strong ionic bonds with any ions of opposite charge that fit close enough to it in the ionic lattice. As you have learned, ions are atoms or molecules bearing an electrical charge. Ionic compounds are made up of positive (cations) and negative ions (anions). In an ionic compound the metal ion doesn’t just form a bond with the ion it donated electrons to.  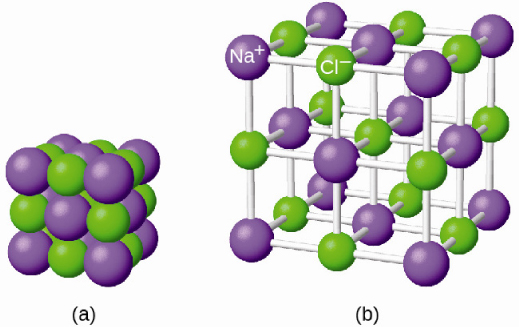
The charge is distributed throughout the ion, the square brackets denote this.You can use this to work out the charge of the ion. The group number on the periodic table tells you how many electrons there are in the outer shell of the atom.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
